A Year in Review 2025
2025 has been a transformational year for Unravel Biosciences, marking its first year as a clinical-stage therapeutics company.
Welcome to our Year in Review.
From the CEO’s Desk
Richard Novak, phdCEO & Co-FounderAs 2025 draws to a close, I want to share my excitement on the remarkable progress our mission-driven team has made in demonstrating the impact of Unravel Biosciences’ AI-driven drug development approach. This year, we reached a transformative milestone by becoming a clinical-stage biotech, just four years after spinning out as a therapeutic discovery company from the Wyss Institute at Harvard University. Our vision for a patient-driven drug development model accelerating the progress to the clinic for the most complex disorders gets closer to reality with each advance at Unravel.
We are thrilled to announce that in January we will begin dosing the first Rett and Pitt Hopkins syndrome patients in Medellin, Colombia. This marks the culmination of years of hard work, many challenges and finding many opportunities as well. Importantly, these clinical programs were guided by families and advocates to ensure that the studies would assess critical quality of life issues that truly impact the patients and caregivers.
These two trials demonstrate that defragmenting rare disease is not only possible but an altogether more efficient process, not only in early discovery stages but also in clinical development. We see this as a turning point for making orphan drug development economically attractive while treating more patients. A win-win!
Beyond the clinic, we launched several biopharma programs, from new target discovery to derisking the complex activities of a new drug candidate. We are employing our BioNAV™ network computation platform, powered by our growing primary patient datamine, creating Living Molecular Twins™ that allow us to simulate the clinical outcomes of 40,000+ molecules in the unique biological state of each patient. This AI-enabled approach using each patient’s molecular data drastically shifts the speed and efficacy of drug development and clinical trials. This resonates with our partners as the current biotech market has pressured biopharma to seek greater efficiency through innovative approaches, while still delivering effective drugs to patients.
To facilitate this growth, we hired four stellar new employees this year, spanning computation, bioinformatics, project management and business development, and expanded our advisory board.
This enabled the growth of our rareSHIFT™ program which provides access to our platform capabilities, such as BioNAV™ and Living Molecular Twins™, to patient advocacy groups, families and clinicians. We are now working with patients across more than 80 disorders across the globe. We are finding that groups of patients could benefit from the same treatments despite having different diagnoses – an opportunity to impact more patients while reducing the number of drugs needing to be developed. We appreciate that even patients with identical mutations may have different symptoms or responses to treatment. In fact, our work with multiple foundations has characterized their very diverse patient populations, optimized their therapeutic translation efforts, aligned drug development from the patient subgroups to preclinical models, and then back to clinical translation.
This alignment, of patient data driven drug discovery through predicting patient translation, inspired a new framework we call Predictable Medicine™. Through Predictable Medicine, we forecast and optimize the entire therapeutic development path: from preclinical models representing specific patient groups, to biomarkers, and to finding patients most likely to benefit, with the lowest chance of adverse events. The goal is better therapies, developed faster, and informed by patient data from the start. rareSHIFT™ has enabled foundations and families to advance to clinical evaluation, with over two dozen therapies being validated in the clinic. We are supporting two foundations by designing observational studies they can quickly use to investigate drugs that could offer a rapid path to updating clinical standard of care for those disorders. Our SquishyWare™ tadpole model data was part of an IND submission to the FDA supporting one of the fastest successfully delivered gene therapy trials. Finally, and despite our results being strictly research data, some clinicians have begun incorporating rareSHIFT™ data as part of their overall treatment strategy.
I want to take a moment to look back at this whirlwind year to appreciate the tremendous partnerships that foundations, families, and other stakeholders have graciously enabled. I am personally grateful to these tireless advocates and partners who placed their trust in us, and who share our belief that patients cannot wait. I also continue to be thankful for the Unravel employees, advisors, and investors who all believe in the power of Predictable Medicine™ and the potential that Unravel offers to patients around the world. As I look forward, 2026 will be a major year for Unravel, including multiple clinical trial readouts, while we continue revolutionizing drug development for families and their patients.
With gratitude,
Richard
A Snapshot of Our Progress
It’s been a busy year for Unravel. Here’s a quick count of some of our work.
Changing the Paradigm of Drug Development: The RVL-001 Clinical Trials
The recent approvals of our RVL-001 clinical trials for Rett syndrome and Pitt Hopkins syndrome have been a major milestone of 2025, with first dosing anticipated in January 2026.
Using our Predictable Medicine™, which combines our datamine of real patient data and our computational AI platform, BioNAV™, we create Living Molecular Twins (LMT) to resemble real patients living with rare disorders. These LMTs enable us to reach the clinic at an accelerated pace by allowing us to understand how real patients will respond to a therapeutic before it ever enters the clinic. We discovered RVL-001 for Rett syndrome in 2019 and Pitt Hopkins syndrome in 2023. With significant gaps in development for financing and starting Unravel, only 2.5 years were needed from discovery to phase I approval.
The clinical trials will be conducted at the Universidad de Antioquia’s Center for Technological Development (“PECET”) in Medellin, Colombia, with Dr. Carolina Lesmes as the Principal Investigator. Thank you to Dr. Lesmes and the PECET team for their hard work and dedication.
Moving into the new year, Unravel will continue pursuing molecules for patient populations with high unmet need using Predictable Medicine™ and our LMTs.
rareSHIFT™
Working Across the World
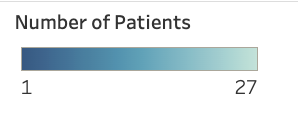
rareSHIFT™ utilizes a non-invasive sampling modality that allows us to work with patients all over the world. Check out all of the places that we’ve been privileged enough to work with patients thus far.
*hover your mouse over the dots to see the number of patients we work with from each location*
Reaching Across Disorders
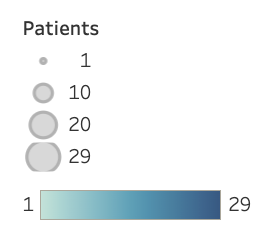
Unravel currently has 83 rare disorder programs. Each gene can be seen to the left, along with the number of patients.
*hover your mouse over the genes to see the number of patients we work with from each disorder*
New Partnerships
In 2025, we partnered with many patients, families, and foundations through rareSHIFT™. This year, we partnered with 18 new foundations.
rareSHIFT™ studies entail making Living Molecular Twins of patient populations based on real patient data. We help families and foundations accelerate their therapeutic odysseys, predicting which existing therapeutics will be the most efficacious, while also indicating which mechanisms should be targeted. Starting in patients enables understanding of how a patient population will respond prior to extensive research into a single drug target that does not have guaranteed success.
Our work with patient populations identifies deep insights about patient populations, often identifying multiple subgroups within a population that respond to therapeutics similarly. Not only do these studies identify the minimum number of molecules needed to treat the maximum number of patients, but they also pave the way for us to defragment rare disease, looking for similar drug responders not just within a disorder but across various disorders, creating larger groups of therapeutic responders to create commercially viable markets for molecules.
In addition, we began a collaboration with COMBINEDBrain to enable large-scale drug discovery for rare neurological disorders. This collaboration will catalyze our goal of defragmenting rare disease. As a consortium for rare neurological disorder advocacy groups, COMBINEDBrain will accelerate its members’ efforts to identify drug candidates and therapeutic mechanisms through the rareSHIFT™ program. Thus, Unravel can identify shared mechanisms to further syndicate our drug development approach and defragment rare disease.
We are grateful for the financial support of all our partners.
Diverse Solutions for Diverse Challenges
BioNAV™’s capabilities aren’t limited to rare disease patients or even to humans. It can tackle a variety of challenges, including biohazards and conservation.
Domoic acid toxicity has been causing increased incidences of sickness and death in marine wildlife due to climate change. In 2024, thousands of sea lions were reported to have domoic acid toxicity across the California coast. Thus, Unravel has partnered with the Zymo Research Corporation and the Channel Islands Marine & Wildlife Institute (CIMWI) to identify treatments for the sea lions.
In 2025, our partnership led to the discovery of a therapeutic candidate to counter neurotoxicity and mortality. CIMWI has conducted critical tests of this molecule in its facilities and is ready for dosing of future stranded animals. In 2026, we are hoping to see the first sea lions rescued by this treatment.
The Unravel Biosciences team extends our gratitude to all of our partners, investors, advocates, advisors, caregivers, and clinicians who have collaborated and supported us this year!